Decentralized Clinical Trials Reduce Burden on Patients
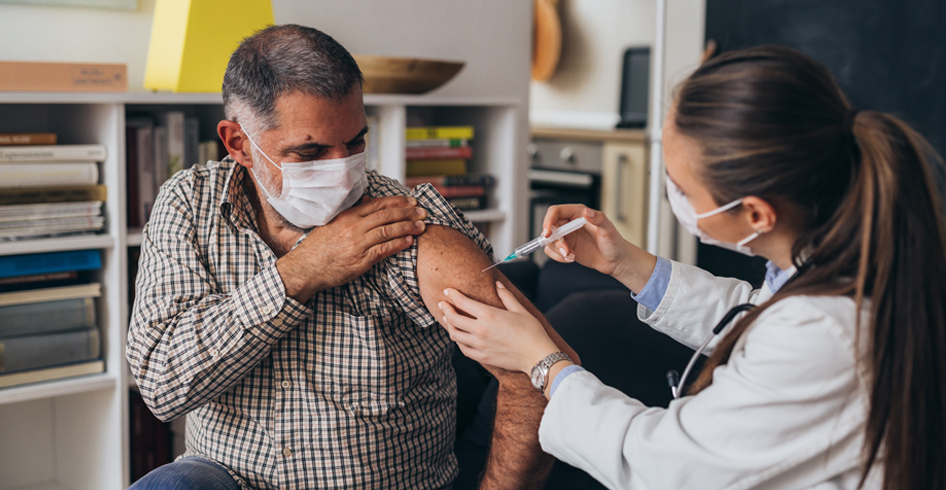
This insightful article by PharmaVOICE discusses how decentralized clinical trials (DCTs) reduce burdens on patients. Nicholas Kenny, Ph.D., chief scientific officer at Syneos Health says, “The key advantage is DCTs allow us to take the trial to the patient as opposed to taking the patient to the site.”
The focus of the story begins by recognizing that COVID-19 catalyzed a monumental shift. Decentralization became a result of the new normal within an industry that had not yet fully embraced the trial model. Driven by the needs of COVID-19, DCTs quickly became a focal point from the onset of the pandemic. But will decentralized trials remain an essential modality going forward? The purpose of the article debates where DCTs will land in a post-COVID clinical trial landscape. Several other leaders provide quotes in the article, including those from PPD, PRA, Parexel, Medidata, Veeva Systems, and many more.
“The message is clear: rather than revert back to traditional clinical trial designs the industry is considering decentralized trial approaches as a valuable asset in the design and execution of future clinical trials,” says Niklas Morton, senior VP, PPD Digital.
The pandemic has also served to illuminate a desired shift on behalf of patient advocacy. And, decentralized trial models provide direct contact with healthcare professionals. “Real success will be achieved when we look at DCTs as a balanced approach to move away from site-centric and toward patient-centric approaches. Otherwise, we run the risk of ostracizing large patient cohorts with fully digital and remote approaches that are not grounded in insights about what will work best for patients,” says April Lewis, executive VP and general manager, Hu.
Lewis goes on to say, “The trend overall is toward patients demanding more control over their treatment journey. DCTs or hybrid trials support these patient needs in ways that traditional clinical trials have not been able to meet. What we are looking at is the democratization of a clinical trial, allowing these often life-saving solutions to be viable care options irrespective of a patient’s zip code or his or her local physician knowing how to connect a patient to a clinical trial.”
At H Clinical, we couldn’t agree more. We will continue to lead decentralized trial support and bring clinical research home for patients across Latin America.

We simplify complex clinical trials across Latin America.
H Clinical simplifies complex clinical trials to meet the needs of all patients and studies. We are the leader in delivering localized, community-based, patient-centric clinical trial services and home healthcare throughout Latin America. At H Clinical, we’re bringing clinical research home.
-
TAMPA - U.S./CORPORATE HEADQUATERS
7901 4th St N, Suite 5706 St. Petersburg, FL 33702 -
MEXICO CITY – CENTRAL AMERICAN HEADQUARTERS
Av. Álvaro Obregón 121, Piso 7 Colonia Roma Norte Cuauhtémoc, Ciudad de México, C.P. 06700 -
SAO PAULO – SOUTH AMERICAN HEADQUARTERS
Rua Salviano Jose Da Silva, No 225 Bairro El Dorado, Sao Jose Dos Campos, SP, Brazil


